Gynecologic Oncology
Latest News
Latest Videos

CME Content
More News

Teenaged girls at average risk don’t need Papanicolaou tests, but some special populations do.

Drs Emily K. Hill, Sangini S. Sheth, and Mark H. Stoler share practice pearls for HPV and cervical cancer screening and express their hope for emerging testing options.
Experts in obstetric and gynecologic management provide insight on the role of HPV genotypes in cervical cancer risk.

Mark H. Stoler, MD, FASCP, provides insight on the role of dual testing in the management of HPV positive women.

Drs Sangini S. Sheth and Mark H. Stoler share their thoughts on the adoption of primary HPV screening and how they foresee changes to cervical cancer screening as more individuals get vaccinated.
Meagan Good shares her scare with uterine cancer to encourage people to spot the early signs of endometrial cancer as part of Eisai and advocacy partners' Spot Her campaign
Spot Her aims to educate people about the signs and risk factors of endometrial cancer, one of the most common yet under-recognized women’s cancers.
Thomas C. Wright Jr., MD; Emily K Hill, MD; and Sangini S. Sheth, MD, MPH, FACOG, comment on the impact COVID-19 had on HPV and cervical cancer screening in their respective practices.
Drs Emily K. Hill, Sangini S. Sheth, and Mark H. Stoler, comment on the implementation considerations surrounding HPV and cervical cancer screening.

Uterine cancer is currently the only cancer type not covered by the World Trade Center (WTC) Health Program.
Experts in obstetric and gynecologic management examine conventional cervical cancer screening strategies and consider their specificity and sensitivity targets.

Drs Mark H. Stoler, Emily K. Hill, Sangini S. Sheth, and Thomas C. Wright comment on the general awareness around HPV and cervical cancer in patients as well as the importance of increasing routine cervical cancer screening approaching those conversations with patients.
Emily K. Hill, MD; Sangini S. Sheth, MD, MPH, FACOG, and Mark. H. Stoler, MD, FASCP, review the rationale of screening for HPV and vaccinating patients to prevent progression to cervical cancer.
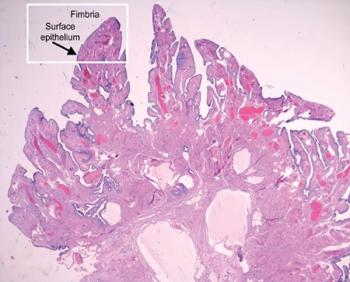
Family history assessment, genetic screening are key to prevention.
Pathology expert Mark H. Stoler, MD, discussed the BD Onclarity™ HPV Assay for primary HPV screening and what it means for the future of cervical cancer detection.
Cancer programs seek to remove barriers to screenings.
A study published in the Journal of Ovarian Research examines factors that may cause endometriosis to progress to ovarian cancer.
Frontline maintenance treatment with selinexor resulted in a statistically significant improvement in median progression-free survival vs placebo in patients with advanced or recurrent endometrial cancer, meeting the primary end point of the phase 3 SIENDO trial.

The campaign emphasizes the importance of annual cancer screenings with the Queen of Hip-Hop Soul
Sarah Lee, MD, MBA, discusses the use of checkpoint inhibitors in endometrial cancer, research regarding the association between MSI-H, dMMR, and TMB-H disease, and the importance of broad molecular testing to ensure all eligible patients are appropriately matched to immunotherapeutic options.

Amer Karam, MD, discusses the role of secondary cytoreductive surgery in ovarian cancer and the need for careful patient selection, plus the results from 3 clinical trials and the key differences between these efforts.
Cortney Eakin, MD, discusses alarming trends in uterine cancer histologies in Black women and some of the potential underlying risk factors at play.

Consensus group discussion resulted in a shift from results-based to risk-based management for cervical cancer screening.
Women are less likely to be diagnosed with bladder cancer, but that does not make the disease any less fatal for this population of patients.
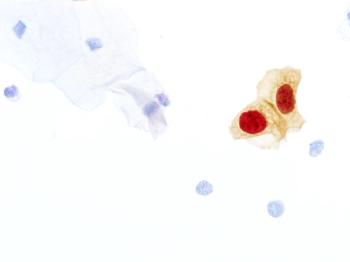
A prospective observational screening study has concluded a cytology dual-stain (DS) biomarker effectively triages women who test positive for primary human papillomavirus (HPV) and are at higher risk of developing cervical cancer, irrespective of the HPV genotype.
Human papillomavirus (HPV) vaccination has significantly decreased the incidence and mortality of cervical cancer in girls and women between the ages of 15 and 24, according to a research letter in JAMA Pediatrics.